 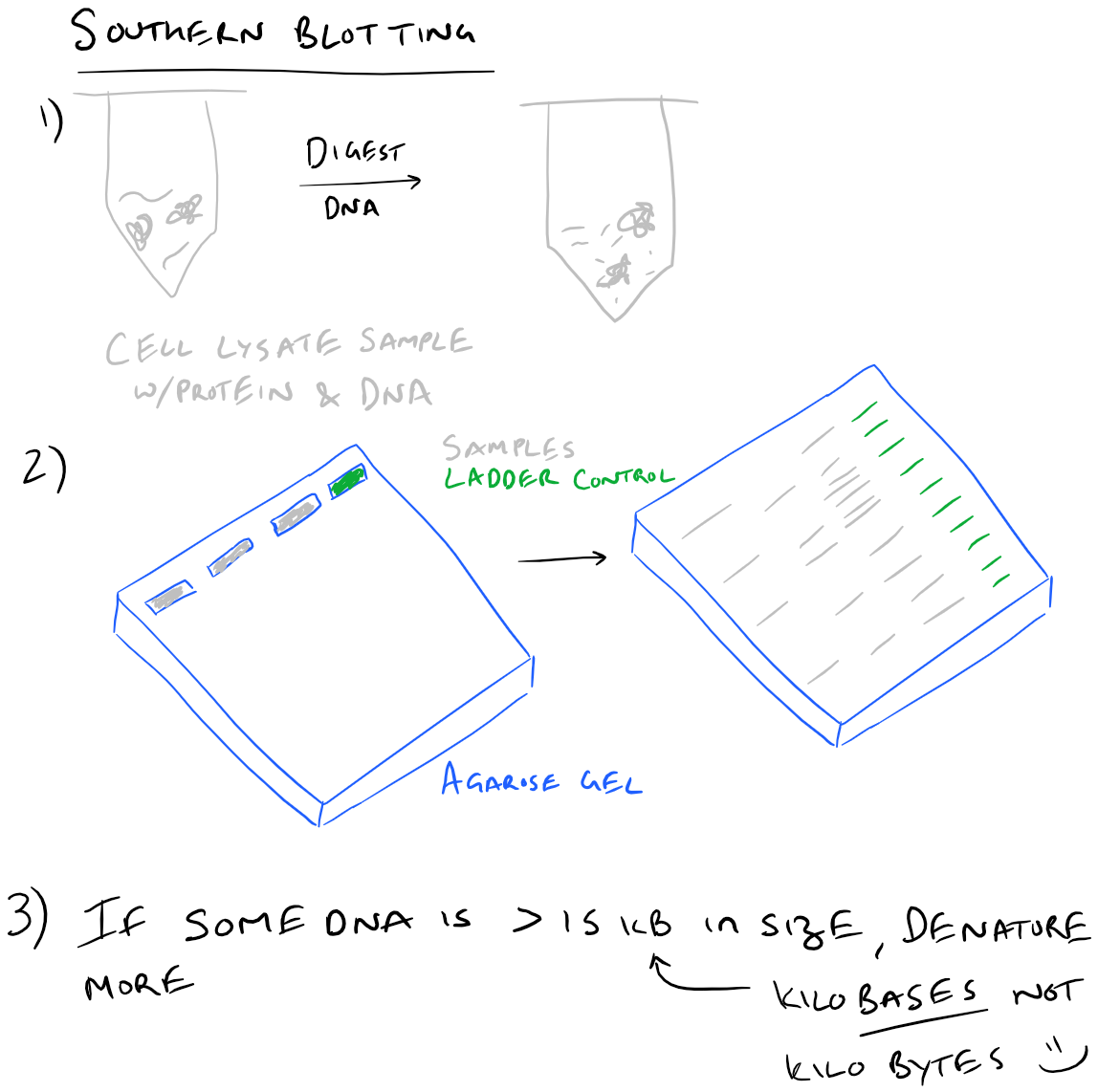
So if the Nowinski lab had been in New York, we would all be doing “eastern” blots.Ī quick aside for the pedants among us. Like nothern blotting, “western blot” was also an allusion to the Southern and nothern techniques, but Burnette had upped the ante by throwing in a geographical reference to location of the Nowinski lab. In a historic, but mostly forgotten conversation with Nowinski, Burnette coined the name “western blot” for his technique. So he developed the method of using electrophoresis to blot the protein onto nitrocellulose paper and after some further work, perfected the technique of blocking non-specific binding sites and visualising the specific radioimmunolabelled antigens using an X-Ray film. Neal Burnette, a post-doc working in the Nowinski group at the Hutchinson Cancer Center in Seattle, started the real fun.īurnette was searching for a way to combine the powers of radio immunoassay and SDS-PAGE electrophoresis so that he could pinpoint specific antigens in a complex protein mixture, such as a cell extract.Īfter some “laughably naive” (his own words – see t his great account by Burnette himself) attempts to visualise the interaction between antibodies and the separated proteins in the gels, he was inspired by Alwine’s nothern blot method (so indirectly by the Southern blot) to make a solid phase replica of the gel. Alwine couldn’t resist the temptation to call his technique the northern blot in an allusion to Southern’s technique, raising a chuckles in labs everywhere. Alwine, a biologist with a sense of humor, developed a technique analogous to the Southern blot, this time for the identification of a specific RNA within a complex RNA sample using a radio-labelled DNA probe. a fractionated genome) and named it after himself – the Southern blot – I’m sure that he had no idea about what he had started. In 1975 when Ed Southern invented his method of using a radiolabeled DNA probe to detect a specific DNA sequence within a DNA sample (e.g. Read on to find out the story of how the Southern, northern and western (etc) blots got their names. It’s a story of discovery, comedy and the triumph of people power over the establishment. This is the story of how one of the most famous and quirky naming conventions in biology came into being. For chemiluminescence signal detection, apparatus need to be disassembled and the membrane need to be taken out and wrapped in a transparent plastic film.It’s official – biologists DO have a sense of humor, well some of them at least. Vacuum-assisted dot blot apparatus has been used to facilitate the rinsing and incubating process by using vacuum to extract the solution from underneath the membrane, which is assembled in between several layers of plates to ensure good seal between sample wells, hold waste solution, and deliver suction force. Finally, for chemiluminescence imaging, the piece of membrane need to be wrapped in a transparent plastic film filled with enzyme substrate. After the protein samples are spotted onto the membrane, the membrane is placed in a plastic container and sequentially incubated in blocking buffer, antibody solutions, or rinsing buffer on shaker. After antibody binding, the membrane is incubated with a chemiluminescent substrate and imaged.ĭot blot is conventionally performed on a piece of nitrocellulose membrane or PVDF membrane. It is then incubated with a primary antibody followed by a detection antibody or a primary antibody conjugated to a detection molecule (commonly HRP or alkaline phosphatase). The membrane is incubated in blocking buffer to prevent non-specific binding. Samples can be in the form of tissue culture supernatants, blood serum, cell extracts, or other preparations. Methods Ī general dot blot protocol involves spotting 1–2 microliters of a samples onto a nitrocellulose or PVDF membrane and letting it air dry. Performing a dot blot is similar in idea to performing a western blot, with the advantage of faster speed and lower cost.ĭot blots are also performed to screen the binding capabilities of an antibody. However, it offers no information on the size of the target protein. The technique offers significant savings in time, as chromatography or gel electrophoresis, and the complex blotting procedures for the gel are not required. Instead, the sample is applied directly on a membrane in a single spot, and the blotting procedure is performed. 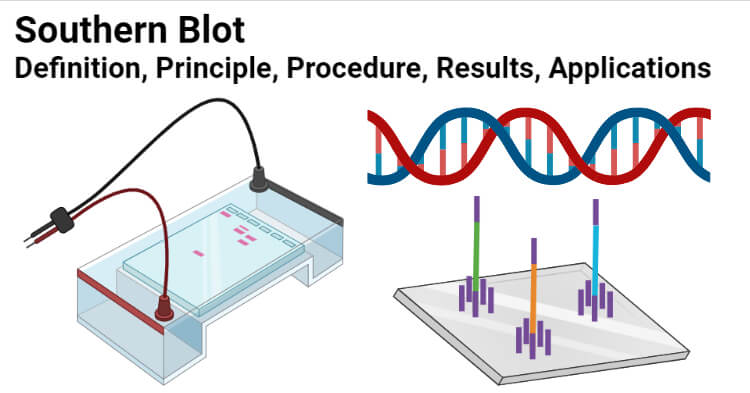 
It represents a simplification of the western blot method, with the exception that the proteins to be detected are not first separated by electrophoresis. Darker dots indicate more protein.Ī dot blot (or slot blot) is a technique in molecular biology used to detect proteins.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
